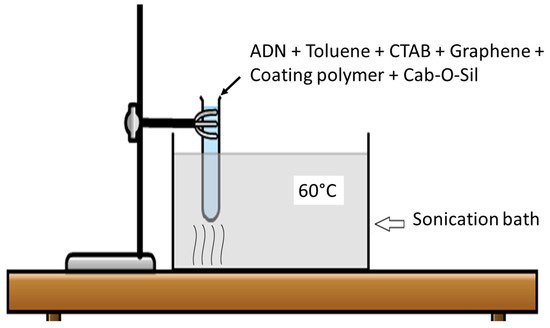
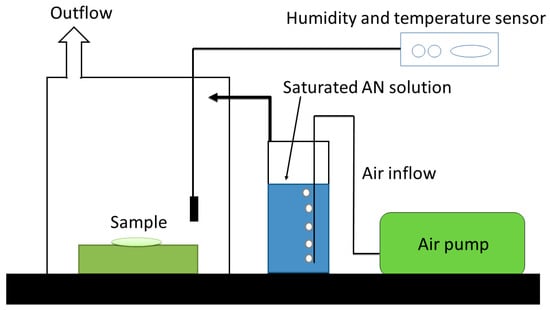
The main objective of these experiments was to increase the density ADN particles by altering the morphology of pristine ADN particles into spherical particles and to add an effective hydrophobic polymer coating. The conventional prilling method involves the melting of ADN. This could pose a potential safety hazard as the energetic ADN particles may decompose during the prilling process. Therefore, the new method based on ultrasound treatment was utilized for alteration of ADN crystal morphology and hydrophobic polymer coating. The main criterion for the ultrasound treatment of ADN was the selection of a suitable sonication medium. First, an organic solvent, which has no solubility toward ADN but has solubility toward the coating polymer, was highly desirable. Secondly, the boiling point of the solvent needed to be sufficiently high to sustain the ultrasound treatment without evaporation. In addition, the solvent needed to be removed easily by vacuum evaporation. This was necessary because excess solvent has to be removed without exposure to atmosphere.
3.1. Selection of Sonication Medium and Polymer
The reduction in particle size is dependent on the surface energy of solids
(σ) [
21]. The interfacial tension
(γ) is the measure of interfacial energy between two immiscible fluids, or solid-fluid, given as [
21]
Equation (1) is termed the Fowkes correlation, where
γ12 is the interfacial tension and
γd1 and
γd2 refer to dispersion forces to the surface tension by liquid 1 and liquid 2, respectively [
21]. In this case, materials 1 and 2 would be the combinations of ADN, toluene, PS, and graphene with each other, as shown in .
Table 2. Interface energy of different components calculated using Equation (2).
However, the value of dispersion forces can be assumed to be 1, giving [
22]
The relation between surface tension and solubility parameter
(δ) is given as [
23]
where V is equivalent to volume.
Furthermore, the interfacial energy is affected by crystal structure of the prilled and coated ADN. The wetting is favored if the surface energy of solid is higher than surface tension of liquid [
23]. The surface tension of ADN melt (
γ=89 mN/m) [
21] is very high due to hydrogen bonding and polarity, compare to water
=72 mN/m, while toluene has a surface tension of
σ=28.4 mN/m. The surface tension of the ADN melt was used to estimate surface energy of the ADN solid.
Based on the bond breaking rule proposed in the previous literature [
24], the surface tension
γliquid(ADN melt) can be correlated to surface energy by the following equation:
Thus, the surface energy of ADN solid is estimated to be σADN=124.8 mN/m.
The difference in surface tension hence favors proper wetting and dispersion of ADN in toluene, which is further enhanced by elevated temperature i.e., 60 °C. The addition of cationic surfactant, CTAB, with an estimated hydrophilic-lipophilic balance (HLB) equivalent number 13+ [
25], failed to disperse the ADN in solvents except toluene. The application of ultrasound sonication facilitates rapid dispersion, which increases the solid-liquid interface.
Using the value of
σADN, the interaction force between different components of the system were calculated as shown below () with data from Krevelen [
23]. In general, the adding of ADN increases the interface energy with ADN-toluence combination that exhibits the highest interface energy.
The PS and graphene adhesion to ADN can be described by the work of adhesion as given by
where γs1 and γs2 are surface energy of materials being considered.
A negative value of
Wadh represents poor adhesion and reversibility of the process.
Wadh values for different components of the system are shown in . They are all in the positive regime. This implies a good adhesion of ADN to PS (
Wadn−ps=144.81 mN/m). Similarly, using the interface energy of
γgr=46.7 mN/m [
26], adhesion of graphene toward ADN and PS was calculated to be
Wadn−gr=169.11mNm and Wgr−ps=88.57mNm, respectively, suggesting a good compatibility among them.
Table 3. Work of adhesion for different components of the system.
3.2. Crystal Morphology and Particle Size
Pristine ADN synthesized in this experiment has long needle-like shape, as shown in , compared to pristine ADN prepared by Heintz et al. [
6]. Spherical ADN particles produced via ultrasound sonication followed by coating with 5% PS and HTPB are shown in . In comparison, ADN melt prilled with conventional method and coated with 5 wt % PS is shown in . The particle diameters were measured from the SEM images using MATLAB.
Figure 3. Scanning electron microscopy (SEM) image of pristine ADN used in these experiments.
Figure 4. (a) SEM image of ADN produced via ultrasound sonication, following by coating with 5% of PS. The particles diameter ranges from 150 µm to 295 µm; (b) SEM image of ADN sonicated and coated with HTPB. The particle diameter ranges from 155 µm to 305 µm.
Figure 5. SEM image of ADN produced by melt prilling and coating with 5 wt % polystyrene (PS).
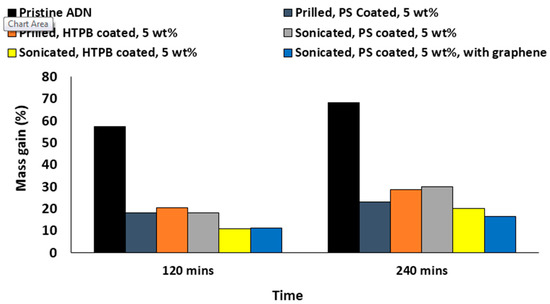
In , the melt prilled ADN particle coating with 5 wt % PS has a size within the range of 173–250 µm. Overall, the ADN particles produced using this method have a mean diameter of 231 ± 48 µm. For ultrasound sonication, the ADN particles have a spherical shape with a particle diameter that ranges from 150 µm to 295 µm, with an overall mean diameter of 225 ± 30 µm. Although the existing ultrasonication set-up could not reduce the mean particle size of the coated ADN particles substantially, it reduced the diameter to the smallest dimension of 150 µm. This is approximately 13% smaller than its counterpart produced using the prilling method. This suggests the ultrosonication method can be optimized further. Nevertheless, the spherical shape of the coated ADN particles produced using the ultrasonication method enables a higher packing density.
3.4. Effect of Additives
Several attempts were made to produce coated ADN without graphene, CTAB, and Cab-o-sil. In the absence of CTAB, approximately 70% of prilled ADN was still left at the bottom of the toluene mixture even after 180 min of sonication. In another attempt to produce coated ADN without graphene and Cab-o-sil, the prilled ADN particles agglomerated in the drying and evaporation steps.
CTAB consists of an anionic long carbon chain (19 carbons) and a relatively weak methylammonium bromide cation, which makes it cationic surfactant [
29].
The cationic part attracts ADN because of the overall negative charge of ADN molecules. Furthermore, the anionic part of CTAB attracts methyl pendant group of toluene by virtue of partial charges leading to proper suspension of ADN in toluene.
In this work, Cab-o-sil was added as a protective colloid to promote emulsification and reduction in viscosity [
30]. Its non-Newtonian behaviour provides better mechanical energy transfer to surface boundary between toluene and AND particles. In addition, it also decreases the agglomeration of particles by inducing steric hindrance [
30]. In this case, the mechanical energy required to reduce the ADN particle size is provided by the ultrasound sonication.
The use of graphene in energetic materials is a rather new idea. It has only been reported for a few propellants with improvements in thermal stability [
31]. However, it has been used extensively to impart hydrophobic properties in polymers, where it acts as a physical barrier or produces a lotus effect [
31] to reduce moisture absorption. As such, graphene was added into the ADN suspension during sonication to reduce moisture absorption. In addition, the adding of graphene is expected to reduce agglomeration of the ADN particles by increasing the distance between ADN particles. shows the SEM image of the agglomeration of ADN particles without the adding of graphene.
Figure 7. Agglomeration of ADN particles.
3.5. Thermal Analysis
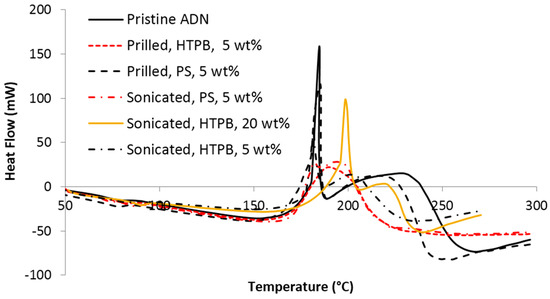
DSC results of pristine, prilled, and sonicated ADN samples are shown in . Pristine and prilled ADN particles were reported to have two peaks in the range of 130 to 220 °C [
32,
33]. The first peak around 130 °C indicates the decomposition of the dinitramide ion to produce ammonium nitrate and N
2O. The second peak, which is minor peak compared to the first, indicates decomposition of ammonium nitrate [
33]. All of our samples exhibit the similar trend of having two peaks. The coating of HTPB and PS on ADN particles has shifted the second peak to the range of 180 to 230 °C. Higher content of HTPB (20 wt %) causes slow burning of ADN sample. Thus, it has the first peak (at 200 °C) in higher temperature as compared to the other peaks. The trend in the DSC result for sonicated ADN particles is similar to the that of pristine and prilled ADN particles. This indicates that the technique used to coat the ADN particles has a negligible effect on the thermal properties of ADN particles. However, an exception was observed for sonicated ADN particles with 20 wt % HTPB. It has higher melting temperature due to the increased amount of HTPB, contributing to the delay in burning. The overall heat release (enthalpy) during decomposition is 1.78–3.35 kJ/g (220–416 kJ/mol), which is in good agreement with published values [
34,
35,
36]. A summary of the results from thermal analysis is tabulated in .
Figure 8. Thermal decomposition in differential scanning calorimetry (DSC) obtained for samples at 10 °C min−1 heating rate.
Table 4. Details of thermal testing.